
Stage IV colorectal adenocarcinoma — cancer history
My Cancer
This page documents the history, biology, and current treatment strategy for my cancer. It is factual and updated as things change.
A note on language: I use clinical terms here because they're precise. If something is unclear, a quick search will get you there faster than me over-explaining it.
Cancer History
Cancer 1 — Thyroid (2019)
My first cancer was papillary thyroid carcinoma, detected after an ultrasound flagged suspicious nodules during a routine checkup. Surgery removed the left lobe of my thyroid. Pathology confirmed a 1.4 cm papillary carcinoma — slow-growing, high survival rate, clean margins. The right lobe remains and has been under regular surveillance since.
That cancer scared me because of the surgery, not the diagnosis. It went well. I went back to work. I filed it away and moved on.
Cancer 2 — Colon (2024)
In May 2024, I went to a Portuguese emergency room with acute abdominal pain. A CT scan and diagnostic procedure identified a mass involving the cecum — the junction where the small intestine meets the colon — along with a perforated appendix. I was treated with antibiotics and discharged for elective surgery.
In June 2024, a robotic-assisted right hemicolectomy removed the right half of my colon along with the primary tumor. The appendix came out with it.
The tumor was classified as Stage IIIC colorectal adenocarcinoma — moderately differentiated, with spread to three nearby lymph nodes and breach of the outer wall of the colon.
Following surgery, I completed 12 cycles of FOLFOX chemotherapy over approximately six months. Treatment intent at that stage was curative. Oxaliplatin — one of the three drugs in FOLFOX — was removed from the regimen during treatment. Remaining cycles were completed on a two-drug regimen.
Recurrence — Peritoneal Carcinomatosis (2025)
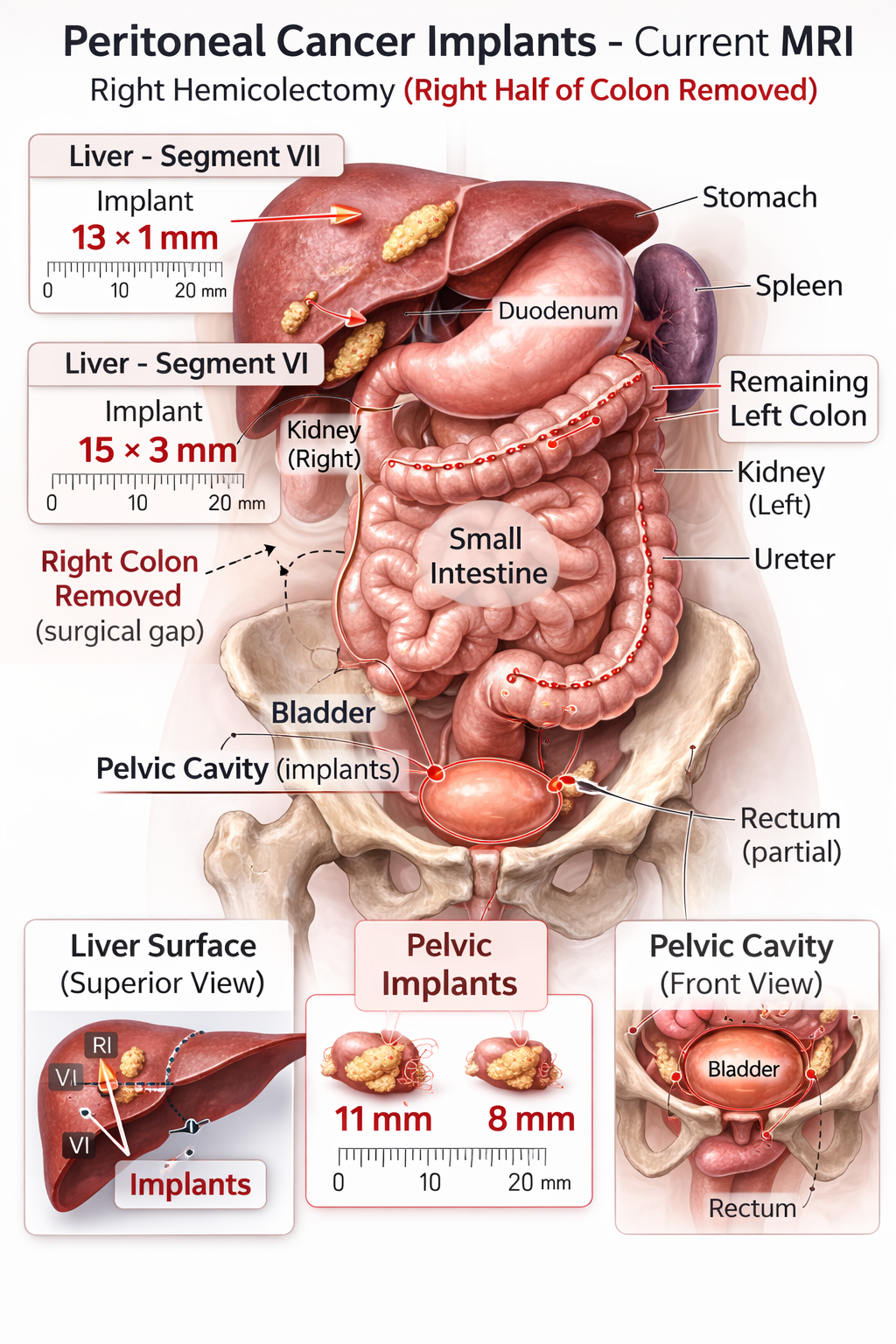
Nine months after completing chemotherapy, follow-up imaging detected cancer again. Small deposits had formed on the peritoneum — the lining of the abdominal cavity — near the liver capsule and in the pelvic region. This is called peritoneal carcinomatosis. It is not the same as liver metastases. The liver itself remains clear.
This reclassified the disease as Stage IV. Second-line chemotherapy with FOLFIRI began in late 2025.
Current Disease Status
- Primary cancer type: Colorectal adenocarcinoma (cecal origin)
- Stage: IV — peritoneal carcinomatosis
- Active sites: Peritoneal implants near the liver (outer surface) and posterior pelvic region
- Current treatment: FOLFIRI — active, with measurable response
Response to Treatment
Early 2026 imaging showed significant improvement. A PET scan showed apparently complete metabolic response — the peritoneal deposits were no longer metabolically active. An MRI confirmed the implants had shrunk substantially; the largest went from approximately 28 mm down to 15 mm. All implants reduced in size. No new lesions appeared.
This is a meaningful response. It does not mean the cancer is gone — it means it is responding to treatment and the current strategy is working.
Imaging Interpretation
Multiple modalities are used to monitor disease. Each measures something different and results should not be conflated.
- PET scan: Measures metabolic activity. A PET-negative result does not mean cancer is absent — it means lesions are not hypermetabolically active at that moment.
- MRI / CT: Measures structural anatomy. Used to track size and location of lesions over time.
Taken together, the current picture is: structurally shrinking, metabolically inactive. That's the best result you can hope for at this stage.
What Comes Next — CRS-HIPEC
Because the peritoneal disease is responding and appears limited in extent, I am being evaluated as a candidate for a procedure called CRS-HIPEC.
- CRS — Cytoreductive Surgery: surgical removal of all visible peritoneal tumor deposits
- HIPEC — Hyperthermic Intraperitoneal Chemotherapy: heated chemotherapy delivered directly into the abdominal cavity during the same operation, targeting any remaining microscopic disease
This is a major surgery — typically six to ten hours, with a recovery of six to eight weeks. It is not offered to everyone. Candidacy depends on the extent of disease, overall health, and response to systemic chemotherapy. The goal is potentially curative — or at minimum, significantly extending disease control beyond what chemotherapy alone can achieve for peritoneal-only disease.
I am currently completing systemic chemotherapy while this evaluation is underway. No surgical date has been set.
Tumor Biology — Genomic Profile (FoundationOne CDx)
Tumor tissue was analyzed using FoundationOne CDx, a comprehensive genomic profiling test that evaluates hundreds of cancer-related genes to identify driver mutations, biomarkers, targeted therapy eligibility, and clinical trial matches.
Key Findings
- KRAS mutation: G12V
- MMR / MSI status: MSS / pMMR (microsatellite stable; mismatch repair proficient)
- Tumor Mutational Burden (TMB): 4 muts/Mb — Low
- HRD status: Negative
- BRAF mutation: None detected
- ERBB2 (HER2) amplification: None detected
Clinical Implications
- KRAS G12V: Predicts resistance to EGFR inhibitors. Cetuximab and panitumumab are not viable options. KRAS G12V-specific drugs exist for a different mutation (G12C) and do not apply here.
- MSS / pMMR + Low TMB: Immunotherapy (checkpoint inhibitors) is unlikely to be effective and is not currently indicated.
- No other actionable mutation: No approved targeted therapy has been identified. Standard chemotherapy remains the primary treatment backbone.
Why Genomic Testing Still Matters
Even without an actionable mutation, comprehensive genomic profiling provides value: it eliminates ineffective therapies, pre-screens for clinical trial eligibility, and establishes a documented molecular baseline. That data is archived and available if the treatment strategy needs to change.
Chemotherapy
Prior Regimen — FOLFOX
- Cycles completed: 12
- Intent: Curative at time of treatment
- Outcome: Disease recurred in the peritoneum approximately nine months after completion
- Dose adjustment: Oxaliplatin was removed from the regimen during treatment. Remaining cycles completed on leucovorin + 5-fluorouracil.
Current Regimen — FOLFIRI
- Schedule: Every 14 days
- Current status: Active — measurable disease response ongoing
- Dose adjustment: Regimen reduced approximately 30% due to elevated liver enzymes.
- Cycle pattern:
- Day 0: ~5+ hour infusion clinic visit, followed by 48-hour portable pump
- Days 1–2: Peak fatigue, nausea, cognitive fog
- Day 3+: Gradual recovery window until next cycle
The Thyroid — Still Watching
The right thyroid lobe — the half that remained after 2019 surgery — showed elevated metabolic activity on a 2026 PET scan. This is a separate finding from the colorectal cancer. The differential includes inflammation from an existing thyroid condition or a new lesion in the remnant tissue.
This is being evaluated. It doesn't change the current colorectal treatment plan, but it adds a layer to the pre-surgical checklist before any major operation moves forward.
Monitoring Plan
- Continue FOLFIRI
- Repeat imaging after several cycles
- Track liver function and inflammatory markers each cycle
- Reassess CRS-HIPEC candidacy based on response and surgical center evaluation
Summary
- Diagnosis: Stage IV colorectal adenocarcinoma
- Key mutation: KRAS G12V
- Targeted therapy options: None currently available
- Immunotherapy: Not indicated (MSS / Low TMB)
- Active treatment: FOLFIRI — responding
- Surgical evaluation: CRS-HIPEC candidacy under assessment
- Genomic data: Archived via FoundationOne CDx for future decisions
Treat. Scan. Adjust. Repeat.